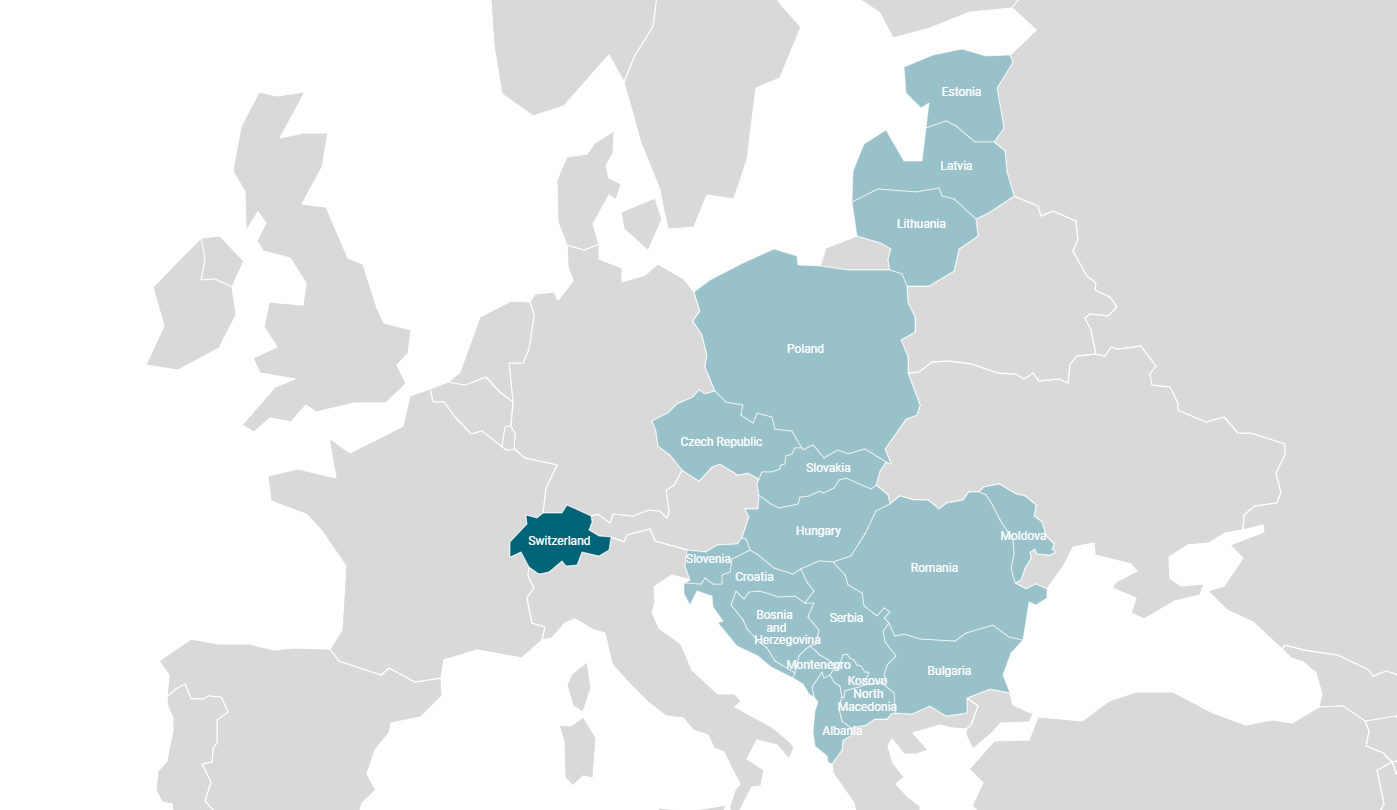
EWOPHARMA – YOUR ENTRY TO CEE
PHARMA MARKETS
At Ewopharma, founded in 1959, we draw on decades of knowledge and experience in the healthcare industry to provide our partner companies with access to the pharmaceutical markets in Central Eastern Europe (CEE) and Switzerland. Our tailor-made medical marketing helps firms in the pharma sector to expand their operations on an international level with a local partner at their side – bringing their healthcare products to a wider audience.